Silicone Breast Implants
Conveniently located to serve the areas of Honolulu, HI
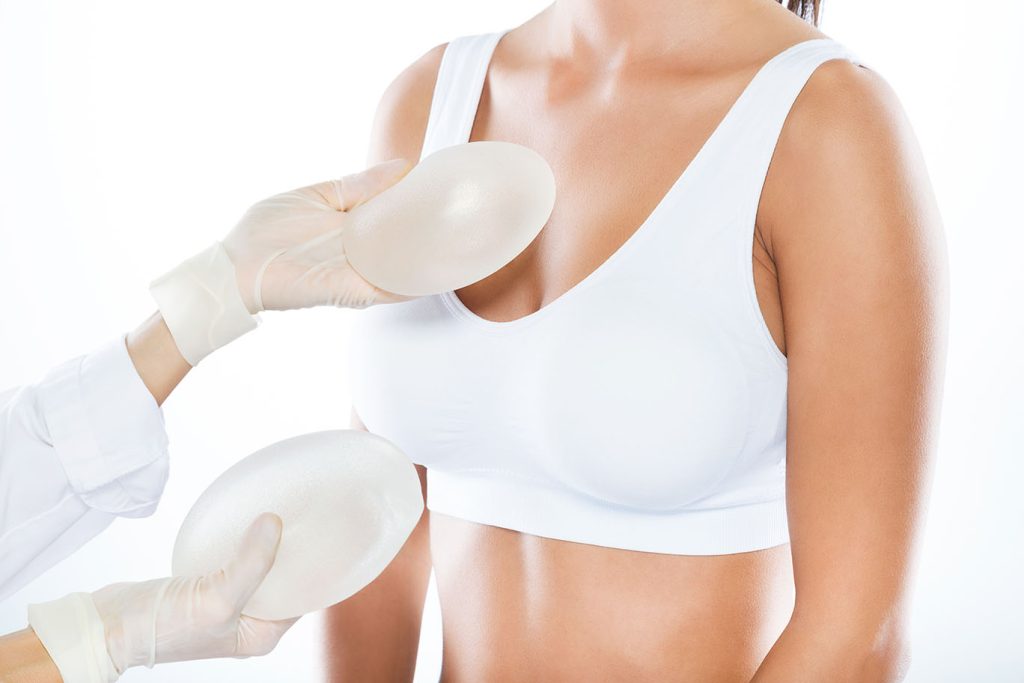
Known for their natural look and feel, silicone implants offer a soft, pliable texture that closely mimics natural breast tissue. They are available in various shapes, sizes, and profiles to suit individual preferences and anatomical considerations. Both Mentor and Inamed make silicone implants with multiple layers of silicone elastomer surrounding the implant. This reduces the amount of gel bleed but can make the implants feel and look firmer. They are, however, much softer than saline.
Contents
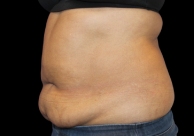
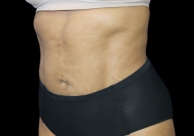
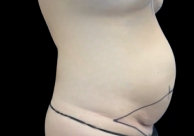
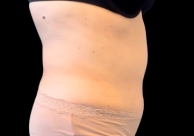
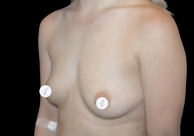
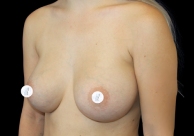
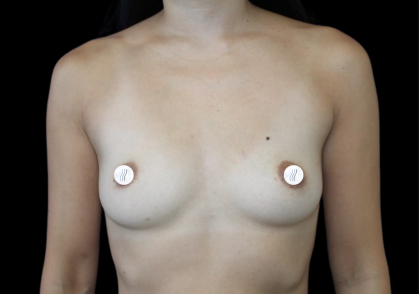
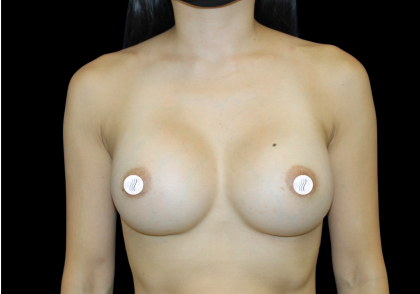
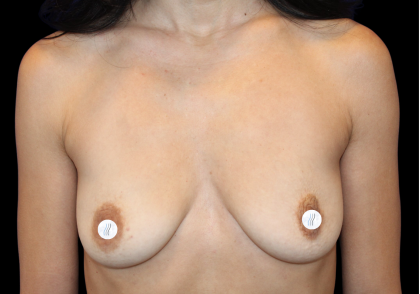
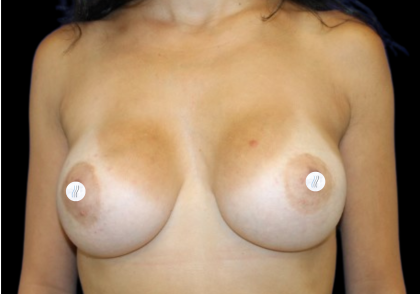
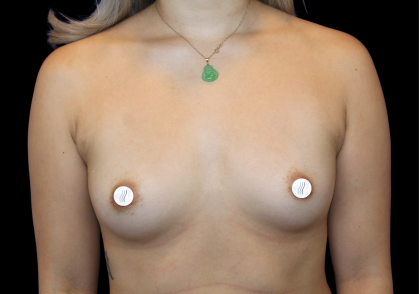
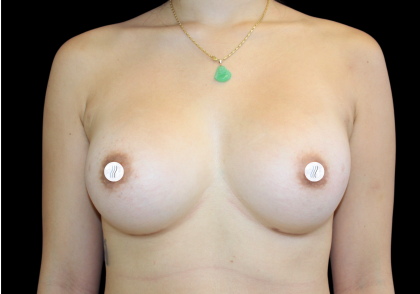
Before and After Photos
Unlike saline breast implants, silicone gel breast implants are pre-filled. Surgeons order them in the requested size and they cannot be changed. Plastic surgeons have to make longer incisions in most cases to place a silicone gel-filled breast implant than a saline-filled breast implant, all other things being equal. The incision for silicone is 4cm compared to Saline which is 3cm, as there is not much difference in the size of the incision this is usually a small part in deciding which implant the patient chooses. Silicone implants cannot be placed endoscopically through a navel incision.
Is Silicone Safe? Studies about Silicone Gel Breast Implants
Numerous studies have been conducted to see if the anecdotal claims of women about their silicone implants were verifiable by scientific evidence. These studies have found localized risks associated with all breast implants, but no conclusive evidence to support the claims against implant manufacturers as noted above. Many studies conducted by the Institute of Medicine and other experts have found no connection between implants and the diseases claimed by so many women.
The FDA has conducted a study where they concluded that extracapsular silicone (from a ruptured implant) can be associated with fibromyalgia. The study relied on a survey of 344 women who filled out surveys asking about their symptoms. Women who reported they had the symptoms were then checked to see if their implants had ruptured. Based on the women reporting symptoms and having ruptured implants, the study assumed a connection. Unfortunately, that is how the plaintiffs prevailed in numerous lawsuits, with no evidence of a cause and effect, just a demonstration of coincidence. Studies have found no link between implants and breast cancer, other than the fact that implants can interfere with detection because of interference with mammography.
Read the Studies about Silicone Gel Breast Implants yourself:
Who Qualifies for Silicone Breast Implants?
According to Mentor’s website, eligibility is as follows: Mentor MemoryGel Silicone Gel-Filled Breast Implants are indicated for females for the following uses (procedures):
- Breast augmentation for women at least 22 years old. Breast augmentation includes primary breast augmentation to increase the breast size, as well as revision surgery to correct or improve the result of a primary breast augmentation surgery.
- Breast reconstruction. Breast reconstruction includes primary reconstruction to replace breast tissue that has been removed due to cancer or trauma or that has failed to develop properly due to a severe breast abnormality. Breast reconstruction also includes revision surgery to correct or improve the result of a primary breast reconstruction surgery. (A separate patient brochure is available for and should be read for breast reconstruction.)
Mentor’s silicone gel-filled breast implants, referred to as MemoryGel products, come in a variety of profiles and sizes. Mentor’s Lumera and Becker gel implants were not approved at this time. Get specific details on the available implant sizes, shapes, etc. in our manufacturer’s section.
What about those “Cohesive Gel” and “Gummy Bear” Silicone Gel Breast Implants?
The current name “gummy bear” is given by many patients and doctors to implants that are “form-stable”. This means that they are SO cohesive, that they are more a solid than a liquid. These form-stable implants have the purpose of making a longer lasting implant that maintains a more attractive and predictable shape. As such, the gel is less liquid-like and more solid-like than even today’s other cohesive silicone gel implants.
What is Silicone? The difference between silicon, silica and silicone.
Silicon is the second most abundant element in the earth’s crust, comprising around 28% of it. It is not found in its elemental form but occurs mainly as oxides and silicates. In contrast to carbon, silicon-silicon bonds are uncommon. Natural silicon-carbon bonds are extremely rare but they can be created synthetically.
Silica is a three-dimensional network of silicon dioxide, most commonly encountered as sand. Silica exists in crystalline and amorphous forms. Silica is chemically resistant at ordinary temperatures but can undergo a variety of transformations at high temperatures (greater than 500ºC) and pressures. The industrial production of amorphous silica requires temperatures of 500ºC and much higher temperatures are required to produce crystalline silica.
The prolonged inhalation of crystalline silica dust is associated with silicosis. Amorphous silica is much less pathogenic than crystalline forms. Conversion of amorphous to crystalline silica cannot occur at body temperature. High-purity amorphous silica is used as a reinforcing agent to increase the tear resistance of silicone rubbers used in medical devices and implants.
Silicones are synthetic polymers and are not therefore found naturally. They have a linear, repeating silicon-oxygen backbone akin to silica. However, organic groups attached directly to the silicon atoms by carbon-silicon bonds prevent formation of the three-dimensional network found in silica. These types of compound are also known as polyorganosiloxanes. Certain organic groups can be used to link two or more of these silicon-oxygen backbones and the nature and extent of this crosslinking enables a wide variety of products to be manufactured. The most important materials used in medical implants are fluids, gels and rubbers (elastomers) whose physical and chemical properties include, amongst others, a high degree of chemical inertness, thermal stability and resistance to oxidation.
Silicone fluids (oils) are usually linear chains of polydimethylsiloxane (PDMS) which have a wide range of chain lengths and molecular masses. Cyclic polydimethylsiloxanes also occur and are important intermediates in the manufacture of linear chain fluids. They are virtually insoluble in water.
Silicone gels have lightly cross-linked polysiloxane networks, swollen with PDMS fluid to produce a cohesive mass. The PDMS fluid is not chemically bound to the crosslinked network but is retained only by physical means, as water is in a sponge, and there is a tendency for the fluid to “bleed”. The degree of cross-linking and amount of fluid affect the physical properties of the gel and the rate at which fluid “bleeds” from it. Once suitably cross-linked, silicone gels retain their form without external containment.
Silicone elastomers are extensively cross-linked and contain little free PDMS fluid. The barrier coating of breast implant shells is a special silicone elastomer that is selected specifically to minimize migration of PDMS from the implants. The tensile strength and tear resistance of silicone elastomers may be increased by the addition of amorphous silica which is usually pre-treated with organosilicon compounds to enable it to be tightly incorporated into the polymer network.